Received 29 September 2017; Revised 29 October 2017; Accepted 3 November 2017; Published 14 December 2017
M. A. Zayed, Arafa Belal, and S. M. A. H. Ragha, Structure Studies of the Prepared Novel Hydrazone Schiff's Base Complexes Using Spectroscopic, Thermal Analyses and Their Biological Activities, Journal of Transition Metal Complexes, 1 (2018), art236047. doi:10.4303/jtmc/236047
The complexing behavior of benzoylhydrazone Schiff's base (A1), with the IUPAC name (2(1-hydrazonoethyl)phenol), towards
the transition metal ions, namely, Mn(II), Fe(III), Co(II), Ni(II), Cu(II), Zn(II),
Hg(II), Ce(IV), and UO2(II), has been examined by elemental analyses, magnetic measurements of some
of them, electronic spectra, FT-IR, 1H-NMR, and 13C-NMR. Thermal properties and
decomposition kinetics of all complexes are investigated. The interpretation of kinetic parameters of thermal
decomposition stages of some of them has been evaluated using Coats-Redfern equation. The antifungal activities of the
free ligand and its metal complexes have been tested in vitro against two types of fungi: Aspergillus fumigatus,
RCMB 02568; Candida albicans, RCMB 05036. Besides, two gram-positive (Bacillus subtilis, RCMB 010067;
Streptococcus pneumoniae, RCMB 010010) and two gram-negative (Escherichia coli, RCMB 010052;
Pseudomonas aeruginosa, RCMB 010043) bacteria were used as test organisms in order to assess the complexes'
antimicrobial potential. It is clear from this survey that the A1 complexes are of high biological activities as
antifungal, in which Cu-A1 and Co-A1 are the most efficient complexes in a level more or less approaches that of
the amphotericin standard. It also indicates that the Cu-A1 and Mn-A1 have the largest biological effects
on gram-positive bacteria of both kinds, and that Cu-A1 complex has a more effect (32.4±0.30) or of the same
order of magnitude (23.8±0.20) as that of the ampicillin standard (23.8±0.2).
hydrazone Schiff's base; spectral studies; transition metal complexes; thermal analyses; biological activity
Schiff's bases have been of good importance in coordination [1] and macrocyclic chemistry [2]. The synthesis
of macrocyclic complexes have great potential applications in fundamental and applied sciences [3,4,5]; biochemistry,
material science, catalysis, encapsulation, activation, transport and separation phenomena, hydrometallurgy, etc. and
importance in the area of inorganic chemistry [6,7]. The development of the work of bioinorganic chemistry has been
another important factor in spurring the growth in interest in macrocyclic compounds [8]. The Schiff-base ligands
with oxygen and nitrogen donor atoms in their structures act as good chelating agents for the transition and nontransition
metal ions [9]. Coordination compounds with metal ions, such as copper, nickel, and iron, often enhance their
activities [3]. There is a continuing interest in metal complexes of Schiff's bases because of the presence of
both hard nitrogen or oxygen donor atoms in the backbones of these ligands. They readily coordinate with a wide range
of transition metal ions yielding stable metal complexes, some of which have been shown to exhibit interesting physical
and chemical properties [5] and potentially useful biological activities [6].
Schiff's bases [6] are still regarded as one of the most potential group of chelators for facile preparations of
metallo-organic hybrid materials. In the past two decades, the properties of Schiff-base metal complexes stimulated
much interest for their noteworthy contributions. These contributions are such as material science [10], catalysis
of many reactions like carboxylation, hydroformulation, oxidation, reduction and epoxidation [11], their
industrial applications [12], and their ability towards some toxic metals [13]. The Schiff-base compounds
are considered highly interesting analytical reagents, because they enable simple and inexpensive determinations of
different organic and inorganic substances [14]. The most important step in the development of metal complexes
was perhaps the preparation of a new ligand, which exhibits unique properties and novel reactivity. Thus, the electron
donor and electron acceptor properties of the ligand, structural functional groups, and the position of the ligand in
the coordination sphere together with the reactivity of coordination compounds may be a factor of different
studies [15,16,17,18,19,20,21,22,23,24,25,26,27]. Schiff's bases are compounds containing an azomethine group
(-CH=N-) which have drawn attention for many years ago. Their metal complexes have been studied, with
a variety of transition metal ions, since they frequently exhibit unusual structural properties [21].
Aspartic proteases are a class of enzymes that play a causative role in numerous diseases such as malaria (plasma epsins),
Alzheimer's disease (β-secretase), fungal infections (secreted aspartic proteases), and hypertension (renin)
[1]. Proteases represent the class of enzymes which occupy a pivotal position with respect to their physiological
roles as well as their commercial applications. They perform both degradative and synthetic functions. Since they are
physiologically necessary for living organisms, proteases occur ubiquitously in a wide diversity of sources such as
plants, animals, and microorganisms. Microbes are an attractive source of proteases owing to the limited space required
for their cultivation and their ready susceptibility to genetic manipulation. Proteases play a critical role in many
physiological and pathophysiological processes. There is a renaissance of interest in using proteolytic enzymes as
targets for developing therapeutic agents [2]. C. albicans is the most common fungal pathogen of humans and
has developed an extensive repertoire of putative virulence mechanisms that allows successful colonization and infection
of the host under suitable predisposing conditions. Extracellular protolytic activity plays a central role in Candida
pathogenicity and is produced by a family of 10 secreted aspartyl proteinases (Sap proteins). Although the consequences
of proteinase secretion during human infections are not precisely known, in vitro, animal, and human studies have
implicated the proteinases in C. albicans virulence in one of the following seven ways: (i) correlation between Sap
production in vitro and Candida virulence, (ii) degradation of human proteins and structural analysis in determining
Sap substrate specificity, (iii) association of Sap production with other virulence processes of C. albicans, (iv) Sap
protein production and Sap immune responses in animal and human infections, (v) Sap gene expression during Candida
infections, (vi) modulation of C. albicans virulence by aspartyl proteinase inhibitors, and (vii) the use of
Sap-disrupted mutants to analyze C. albicans virulence. Sap proteins fulfill a number of specialized functions
during the infective process, which include the simple role of digesting molecules for nutrient acquisition,
digesting or distorting host cell membranes to facilitate adhesion and tissue invasion, and digesting cells
and molecules of the host immune system to avoid or resist antimicrobial attack by the host [3].
The present study describes the chelation behavior of Schiff's base (A1), derived from the condensation of 2-hydroxy
acetophenone with hydrazine hydrate, towards some transition elements; which may help in more understanding of the mode
of chelation towards metals. For this purpose, the complexes of Mn(II), Fe(III), Co(II), Ni(II),
Cu(II), Zn(II), Hg(II), Ce(IV), and UO2(II) ions with H2L are studied in solution and
in solid states. The structures of the studied complexes are elucidated using elemental analyses, FT-IR, 1H-NMR,
solid reflectance, magnetic moment, molar conductance, and thermal analyses (TGA, DTGA, and DTA) measurements. The
biological activity of the parent Schiff base and its metal complexes had been practically and theoretically studied.
All chemicals used in this study were of the analytical reagent grade and of highest purity available. They
included Cu(II) chloride (Sigma-Aldrich, MO, USA); Co(II) and Ni(II) chloride hexahydrates
(BDH Middle East LLC, Dubai, UAE); ferric chloride hexahydrate (BDH Prolabo Chemicals, Lyndhurst, South Africa);
Zn(II) chloride (Ubichem, Worcestershire, UK); Hg(II) chloride, Ce(IV) sulphate, and UO2(II)
nitrate (Sigma). Organic solvents used included absolute ethyl alcohol and dimethylformamide (DMF). These solvents
were spectroscopic pure from BDH. Distilled water collected from all glass equipments was used in all preparations.
The metal complexes of A1 were prepared by the addition of hot solution (60 ℃) of the appropriate metal ion
in absolute ethanol (15 mL) to the hot solution (60 ℃) of the organic ligand (0.3 g A1) in ethanol and DMF (15 mL). The resulting mixture was heated with stirring to evaporate all the solvents to get precipitate. The
purified precipitates by washing with hot ethanol-water mixture (1:1) were dried and weighed to calculate the yield.
All the above steps were repeated for all the selected transition metal complexes.
The molar conductance of metal complexes solutions in DMF (10−3M) was measured using Sybron-Barnstead conductometer
(Meter-PM, E = 3,406). Elemental microanalyses of the separated solid chelates for C, H, N, and S were performed in the
microanalytical center at Cairo University. The analyses were repeated twice to check the accuracy of the data.
Infrared spectra were recorded on a Perkin-Elmer FT-IR type 1650 spectrophotometer in the region
4,000–400 cm−1 as KBr disks. The 1H-NMR spectra were recorded with a JEOL EX-270 MHz
in DMSO-d6 as solvent, where the chemical shifts were determined relative to the solvent peaks. The diffused reflectance
spectra of some of these complexes were measured on a Shimadzu 3101 PC spectrophotometer. The molar magnetic
susceptibility was measured on powdered samples using Faraday method. The diamagnetic corrections were made
by Pascal's constant and Hg[Co(SCN)4] was used as a calibrant. The magnetic data for the background of
the sample holder were corrected. The thermal analyses (TG, DTG, and DTA) were carried out in dynamic nitrogen
atmosphere (20 mL min−1) with a heating rate of 10 ℃ min−1
using Shimadzu TG-60H and DTA-60H thermal analyzers.
The tests were done in the Regional Center for Mycology and Biotechnology (RCMB) Culture Collection. These tests were
done using the diffusion agar technique. Spore suspension (0.5 mL, 10−6–10−7 spore mL−1)
of each of the investigated organisms was added to a sterile agar medium just before solidification, then poured into
sterile Petri dishes (6 cm in diameter) and left to solidify. Using sterile cork borer (6 mm in diameter),
three holes (wells) were made into each dish, and then 0.1 mL of the test compound dissolved in DMF
(100 mg mL−1) was poured into these holes. The dishes were incubated at 37 ℃
for 48 h where clear or inhibition zones were detected around each hole. DMF (0.1 mL) was used as a control
under the same conditions. By subtracting the diameter of the inhibition zone resulting with DMF from that obtained
from each metal complex or the free Schiff base, antibacterial activities were calculated as a mean of three replicates.
MIC50 was determined and was defined as the lowest compound concentration that inhibits growth by 50%.
Docking study was performed using the MOE 2014.09 software. Downloading the crystal structure of the secreted aspartic
proteinase 2 (Sap2) PDB ID (1EAG) was carried out from protein data bank website. Regularization and optimization
for protein and ligand were performed. Determination of the essential amino acids in binding site was carried out
and compared with that present in literature. The performance of the docking method was evaluated by redocking
crystal ligand into the assigned active Sap2 to determine the root-mean-square deviation (RMSD) value. Interactive docking
was carried out for all the conformers of interesting compound to the selected active site. Each docked
compound was assigned a score according to its fit in the ligand binding pocket (LBP) and its binding mode.
Optimization of the ADME (absorption, distribution, metabolism, and excretion) properties of the drug molecule is often
the most difficult and challenging part of the whole drug discovery process. The ADME profile will also have a major impact
on the likelihood of success of a drug. Keeping an eye of molecular properties throughout the drug discovery
process is a critical aspect of the process, with the focus at different stages. The hunt for new drugs can be divided
into two stages: discovery and development. Drug discovery includes generating a hypothesis of the target receptor for
a particular disorder and screening the in vitro and/or in vivo biological activities of the new drug candidates. Drug
development involves the assessment of efficacy and toxicity of the new drug candidates.
To aid in a discovery program, accurate data on pharmacokinetics and metabolism must be available as early as possible
as it eventually contributes to the final success or failure of the compound. The initiation of early absorption,
distribution, metabolism, and excretion (ADME) screening has dramatically decreased the proportion of compounds failing
in clinical trials. The main aim of preclinical ADME is to eliminate weak drug candidates in the early stages of drug
development which allow resources to be focused on potential drug candidates. Undesirable pharmacokinetic properties,
such as poor absorption, too long or too short half-life (t1/2), and extensive first-pass metabolism, majorly contribute
to the failure of many drug candidates in early stages of drug development programs. To be successful, a drug candidate
needs to possess good bioavailability and a desirable half-life (t1/2).
The ligand under investigation (A1) is a novel Schiff base; it has a general formula (C8H10N2O),
mole mass = 150, and a structural formula (Figure 1).

Figure 1: Stereo structure of A1 and its numbering system.
Metal complexes of this novel Schiff base were obtained through the reaction between metal ions and H2L ligand at 1:2 or 1:3 (M:L) ratio. The synthesized Schiff-base ligand and its complexes are very stable at room
temperature. The metal complexes are commonly soluble in DMF and dimethyl sulfoxide (DMSO). The elemental analyses,
color, and melting points of metal complexes are presented in Table 1.

Table 1: The analytical data of the proposed stoichiometry of the complexes.
3.1.1. 1H-NMR and 13C-NMR
The HNMR spectra of ligand previously published [23] show a sharp band at 8.4 ppm and 8.5 ppm which may be
assigned to the protons of two azomethine groups (-CH=N-), respectively. These signals are completely shifted
in the Zn complex (Figure 2), indicating that azomethine groups are shared in chelation process. However, a
single and multiple bands at 4.9 ppm and 6.4–8 ppm attributed to methylene and the aromatic protons in the free A1
ligand are shifted to 4.3 ppm and 6.2–7.6 ppm, respectively, which indicates the coordination of ligand to Zn(II) ion.
13C-NMR refers to the appeal of signal of chemical shift 157.2 of C-OH of phenolic group and the signal
at 143.3 of -CH=N- in the free ligand A1. These signals are shifted to other values of chemical shifts: 168.8 and 158.3;
which may refer to the sharing of these groups in complex formation.

Figure 2: The 1H-NMR of Zn(II)-A1-complex.
3.1.2. Thermal behavior of A1-metal complexes and thermodynamic calculations
Thermal analyses' results of the investigated Schiff-base metal (II)/(III) complexes are given in Table 2.

Table 2: TG and DTG analysis of A1-complexes.
3.1.3. Molar conductance measurements
Conductivity measurements in nonaqueous solutions have been used in structural studies of metal chelates within the
limits of their solubility. They provide a method of testing the degree of ionization of the complexes: the molar ions
that a complex liberates in solution, the higher will be its molar conductivity and vice versa. The molar conductivities
of 10−3 molar solutions of the metal chelates at 25±2 ℃ are measured. The molar conductance values
(110–125 Ω−1 mol−1 cm2) of the Co(II), Ni(II),
Cu(II), and Zn(II) complexes with A1 ligand are considered as 1:2 electrolytes as confirmed by the given
proposed general formulae of these complexes. In addition, Fe(III) and Ce(IV)-A1 complexes have molar
conductance value of 250–266 Ω−1 mol−1 cm2, indicating
their ionic nature and they are considered as 1:3 electrolytes [22].
3.1.4. IR spectral studies
The IR spectra of the complexes are compared with the free ligand [23] in order to determine the coordination sites
that may involve in chelation. The spectrum of free ligand shows a band in the region 1637 cm−1
characteristics of the azomethine stretching mode. This band was shifted towards lower frequencies in the spectra of its
metal complexes (1605–1623 cm−1) (Table 3). This phenomenon appears to be due to the
coordination of azomethine nitrogen to the metal ion [23,24]. New bands are found in the spectra of complexes in the
regions 608–648 and 498–601 cm−1, which are assigned to (M–O), (M–N), respectively, and stretching
vibrations. Therefore, from the IR spectra it is concluded that A1 Schiff's base is tetra dentate ligand with
NNOO coordination sites with one or 2H2O molecules of crystallization present in the moiety of some complexes. Therefore, from these data the proposed structural formulae for these complexes can be given by Figure 3.

Table 3: The FT-IR spectra of H2L (A1) and its metal complexes.
3.1.5. Magnetic susceptibility and electronic spectral studies
The diffused reflectance spectral data of the complexes, position of bands, and their transitions assignments are of
particular importance as they are highly dependent on the geometry of the molecule. From the diffused reflectance
spectrum of the Fe(III) complex, the bands at 14,096 cm−1, 18,625 cm−1, and 24,585 cm−1 may
be assigned to the 6A1g→T2g (G) and 6A1g→5T1g transitions in octahedral geometry of the
complex [25]. The spectrum shows also a band at 27,777 cm−1 which may be attributed
to ligand to metal charge transfer. This geometry is further supported by its magnetic susceptibility value (3.8 BM) [25,26,27].
The diffused reflectance spectrum of the Cu(II) complex displayed the d-d transition band in the region
13,606 cm−1 which is due to 2Eg→2T2g transition. This d–d transition band
strongly favors a distorted octahedral geometry around the metal ion. This geometry is further supported by its
magnetic susceptibility value (1.5 BM) [25,26,27].
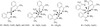
Figure 3: The proposed structural formulas of A1 complexes.
The spectrum of Ni(II) complex displayed three d-d bands at 13,150 cm−1, 14,749 cm−1, and 23,310 cm−1.
These bands correspond to 3A2g (F)→3T2g (F), 3A2g (F)→3T1g (F), and 3A2g (F)→3T1g (P)
transitions, respectively, being characteristic of an octahedral geometry. This geometry is further supported by its
magnetic susceptibility value (2.4 BM) [25,26,27].
Diffused reflectance spectrum of Co(II) complex displayed the d-d transition bands in the regions 15,017 cm−1,
16,723 cm−1, and 23,566 cm−1 which are assigned to 4T1g (F)→4A2g (F), 4T1g (F)→4T2g (F),
and 4T1g (F)→4T1g (P) transitions, respectively. The transitions correspond to the octahedral geometry of
the complex which is also supported by its magnetic susceptibility value (3.06 BM) [25,26,27,28].
The proposed structural formulae of the given complexes depending upon the above data and discussion are given
in Figure 3.
Schiff's bases are important class of compounds in medicinal and pharmaceutical fields.
They show biological applications
including antibacterial [29,30,31,32,33] and antitumor activities. Microbes encounter a variety of metal ions in
the environment and interact with them, which is sometimes beneficial or detrimental depending on the chemical/physical
nature and oxidation state of the metal ion. The microbes have the ability to bind to metal ions present in the external
environment at the cell surface and to transport them into the cell for various intracellular functions. All microbes,
prokaryotes or eukaryotes, were employed metal species for structural and/or catalytic functions. Antibacterial activity
of the Schiff base and its metal complexes were tested by diffusion agar method [29,30,31,32,33]. Amphotericin B was used as a reference for antifungal; ampicillin and gentamicin were used as a reference compounds for
antibacterial activities. In testing the antibacterial activity of these compounds, more than one test organism was used
to increase the chance of detecting antibiotic principles in tested materials. Two types of fungi
were used (Aspergillus fumigatus (RCMB 02568) and C. albicans (RCMB 05036)); also two
gram-positive (Bacillus subtilis (RCMB 010067) and Streptococcus pneumoniae (RCMB 010010)) and two
gram-negative (Escherichia coli RCMB 010052 and Pseudomonas aeruginosa RCMB 010043) bacteria
were used as test organisms. The data are expressed in the form of mean ±SD and minimum inhibitory zone (MIC).
Mean zone of inhibition in mm ± standard deviation (SD) beyond well diameter (6 mm) is produced on a range of
clinically pathogenic microorganisms using (5 mg/mL) concentration of tested sample. The data are depicted
in Table 4.

Table 4: Biological activity of Schiff's base A1 and its metal complexes.
From these data it is clear that the standard amphotericin B has mean zone of inhibition ±SD = 23.7±0.1
against the fungus A. fumigatus (RCMB 02568); while the biological effects of complexes are ordered as Cu-A1
(20.2±0.55) > Co-A1 (18.7±0.36) > Zn-A1 (17.6±0.58) > Cr-A1 (16.8±0.39) > Ce A1 (15.7±0.36)
> Ni-A1 (15.7±0.33) > Mn-A1 (15.3±0.55) > Hg-A1 (13.6±0.25) > Fe-A1 (12.6±0.25). From these
data it is also clear that the standard amphotericin B has mean zone of inhibition ±SD = 25.4±0.1 against the
fungus C. albicans (RCMB 05036); while the biological effects of complexes are ordered as Cu-A1 (19.6±0.33)
> Co-A1 (16.9±0.27) > Cr-A1 (15.9±0.44) > Zn-A1 (15.4±0.25) > Ni-A1 (13.8±0.25) > Mn-A1
(13.4±0.35) > Ce-A1 (13.3±0.36) > Hg-A1 (11.7±0.34) > Fe-A1 (11.2±0.33). This survey shows
that the A1-complexes are of high biological activities as antifungal; in which Cu-A1 and Co-A1 are the most
efficient complexes in a level more or less approaches that of standard.
The biological activity of A1 complexes against gram-positive bacteria (B. subtilis (RCMB 010067)
and S. pneumoniae (RCMB 010010)) refers to the fact that Fe-A1 and Ce-A1 complexes have nondetected (ND)
effects. The standard ampicillin has a zone effect of 23.8 ± 0.2 on S. pneumoniae
(RCMB 010010). The other complexes' effects on S. pneumoniae (RCMB 010010) are ordered in their
increasing effect: Cu-A1 (23.8±0.20) > Mn-A1 (17.5±0.44) > Ni-A1 (16.9±0.58) > Cr-A1
(16.7±0.36) > Hg-A1 (14.6±0.58) > Co-A1 (12.9±0.63) > Zn-A1 (12.3±0.58). The standard
ampicillin has a zone effect of 32.4±0.4 on B. subtilis (RCMB 010067). The A1 complexes'
effects on B. subtilis (RCMB 010067) are ordered in their increasing effect: Cu-A1 (32.4±0.30) >
Mn-A1 (19.8±0.63) > Cr-A1 (19.2±0.27) > Ni-A1 (18.2±0.44) > Hg-A1 (14.3±0.58) > Co-A1
(13.2±0.58) > Zn-A1 (12.7±0.37). It is clear from these data that the Cu-A1 and Mn-A1 have the largest
biological effects on gram-positive bacteria of both kinds; and Cu-A1 complex has a more effect (32.4±0.30)
or of the same order of magnitude (23.8±0.20) of the ampicillin standard (23.8±0.2).
The biological activity of A1 complexes on gram-negative bacteria (E. coli (RCMB 010052) and
P. aeruginosa (RCMB 010043)) refers to the fact that only copper complex has an effect of
17.3±0.1 on P. aeruginosa (RCMB 010043) which of the same magnitude of the standard
gentamicin having an effect of (17.3±0.1). The other complexes have ND effects on
P. aeruginosa (RCMB 010043) and both Fe and Ce complexes also have ND effects on
E. coli (RCMB 010052). The effects of other complexes on negative bacteria E. coli
(RCMB 010052) are in the order Cu-A1 (19.9±0.30) > Mn-A1 (18.9±0.25) > Cr-A1 (13.6±0.36) >
Ni-A1 (11.9±0.63) > Co-A1 (10.8±0.44) > Hg-A1 (9.4±0.44) > Zn-A1 (8.5±0.37).
Molecular modeling was done using MOE 2014.09 software package in reckoning or analyzing all parameters to the docked
amount or score that might also have a prescribed relationship according to the aspartic proteases inhibition
[34,35,36,37,38,39]. The results obtained of ligand A1 are given by Table 5 and Figure 4.

Table 5: Ligand interactions report.
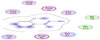
Figure 4: Two dimensional diagram of ligand A1 interactions with the targeting enzymes.
These results indicate that the parent ligand A1 has no binding energy and it has no affinity for binding with aspartic
proteases or any other enzyme of organisms and consequently no inhibition effect to these organisms.
Figures 5–7 show an interaction between the Cu-A1 complex with different Candida enzymes.
Figure 5 shows two-dimensional representations showing the hydrogen bond interactions
of the interesting compound and the targeting enzyme.
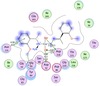
Figure 5: Possible hydrogen bonding between complex A1 and different enzymes of organism.
Figure 6 shows three-dimensional representations of interesting complex pose fitted into the binding
pocket of the targeting enzyme.
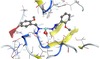
Figure 6: Possible binding of complex A1 with interest organism enzyme.
Figure 7 shows a fitting of the complex molecule into the targeting enzyme's pocket, referring to
alternating its conformation and making inhibition to the organisms under test.

Figure 7: Complex A1 fitted into the binding pocket of the targeting enzyme.
To be successful, a drug candidate needs to possess good bioavailability and a desirable half-life (t1/2). Early
ADME provides the necessary data for selecting preclinical candidates, appropriate dosage forms, and formulation. Also,
it accelerates the timeline for investigational new drug applications and subsequently new drug application submission
to the FDA(4); see Table 6.

Table 6: Bioactivity and ADME toxicity.
The careful survey of this research leads to the following important conclusions:
- (1) preparation, structure investigation, and biological importance of a novel Schiff base (A1) and its metal
complexes of Fe(III), Co(II), Ni(II), Cu(II), Mn(II), Ce(IV), UO2(II),
Hg(II), and Zn(II) are given. It involves elemental, spectroscopic (IR, NMR, and mass spectra), and thermal
data (TG and DTA). It gives a structure confirmation of these complexes;
- (2) thermal properties and thermal decomposition possibilities of all complexes are investigated and have been
evaluated. This study refers to their thermal stability; and this stability may be related the coordination power of
metal cation to the remainder part;
- (3) it is found that the Schiff-base ligand (A1) and its complexes have an important biological effect on two types of fungi
(A. fumigatus (RCMB 02568) and C. albicans (RCMB 05036)); two
gram-positive (B. subtilis (RCMB 010067) and S. pneumoniae (RCMB 010010)) and two
gram-negative (E. coli (RCMB 010052) and P. aeruginosa (RCMB 010043)).
- (4) the A1 complexes are of high biological activities as antifungal, in which Cu-A1 and Co-A1 approached that
of standard;
- (5) it is clear from these data that the Cu-A1 and Mn-A1 have the largest biological effects on
gram-positive bacteria of both kinds; and Cu-A1 complex has a more effect (32.4±0.30) or of the
same order of magnitude (23.8±0.20) of the ampicillin standard (23.8±0.2);
- (6) the data obtained refers to the fact that only copper complex has an effect of 17.3±0.1
on P. aeruginosa (RCMB 010043) which of the same magnitude of the standard gentamicin
has effect of (17.3±0.1);
- (7) the other complexes have ND effects on P. aeruginosa (RCMB 010043)
and both Fe and Ce complexes also have ND effects on E. coli (RCMB 010052).
This research was supported by the Chemistry Departments at both Cairo and Port Said Universities.
Thanks also are presented to the staff at Cairo University for conducting the microanalyses.
The authors declare that they have no conflict of interest.
- R. M. Tada, T. S. Maheta, M. B. Gondaliya, and M. K. Shah, Synthesis and characterization of Cu(II), Ni(II) and Co(II) based 1, 4-substituted thosemicarbazone complexes, Chem Sci Trans, 2 (2013), 135‒140.
- N. Manav, N. Gandhi, and N. K. Kaushik, Some tribenzyl Tin(IV) complexes with thiohydrazides and thiodiamines. Synthesis, characterization and thermal studies, J Therm Anal Calorim, 61 (2000), 127‒134.
- A. K. Mishra, S. B. Mishra, N. Manav, and N. K. Kaushik, Thermal and spectral studies of palladium(II) complexes, J Therm Anal Calorim, 90 (2007), 509‒515.
- M. N. Patel, P. B. Pansuriya, and M. R. Chhasatia, Synthesis, spectroscopy, thermal and biological aspect of novel six-coordinated dimeric iron(III) mixed-ligand complexes, Appl Organomet Chem, 22 (2008), 415‒426.
- M. X. Li, J. Zhou, C. L. Chen, and J. P. Wang, Synthesis, crystal structure and antitumor study of a zinc complex of the 2-benzoylpyridine thiosemicarbazone ligand, Z. Naturforsch, 63b (2008), 280‒284.
- P. Kumar, Synthesis and antimicrobial activities of the 4-chloro-N′-(4-methoxybenzylidene)-benzohydrazide Schiff base promoted by metal(II) ions, The Experiment, 11 (2013), 476‒482.
- J. Kaizer, Z. Zsigmond, I. Ganszky, G. Speier, M. Giorgi, and M. Réglier, New functional model complexes of intradiol-cleaving catechol dioxygenases: properties and reactivity of CuII(L)(O2Ncat), Inorg Chem, 46 (2007), 4660‒4666.
- W. Kaim, C. Titze, T. Schurr, M. Sieger, M. Lawson, J. Jordanov, et al., Reactivity of copper(I) complexes with tripodal ligands towards O2: structures of a precursor [L3CuI(NCCH3)](BF4), L3 = tris(3-isopropyl-4,5-trimethylenepyrazolyl)methane and of its oxidation product [L3CuII(μ-OH)2CuIIL3](BF4)2 with strong antiferromagnetic spin-spin coupling, Z Anorg Allg Chem, 631 (2005), 2568‒2574.
- M. G. Abd El Wahed, E. M. Nour, S. Teleb, and S. Fahim, Thermodynamic and thermal investigation of Co(II), Ni(II) and Cu(II) complexes with adenine, J Therm Anal Calorim, 76 (2004), 343‒348.
- M. M. Taqui Khan, S. B. Halligudi, S. Shukla, and Z. A. Shaikh, Reductive carbonylation of nitrobenzene to phenylurethane catalyzed by Ru(III)-Schiff base complexes, J Mol Catal, 57 (1990), 301‒305.
- U. Singh, R. Singh, W. Devi, and C. Singh, Schiff base complexes of copper(II) ions: synthesis, characterization and antimicrobial studies, J Chem Pharm Res, 4 (2012), 1130‒1135.
- M. M. Deshpande, I. H. Seema, and P. A. Kulkarni, Preparation, physical characterization and antimicrobial evaluation of Co(II), Ni(II) and Fe(III) complexes of heterocyclic Schiff bases, Int J Biol Pharm Res, 4 (2013), 460‒464.
- G. Kumar, D. Kumar, C. P. Singh, A. Kumar, and V. B. Rana, Synthesis, physical characterization and antimicrobial activity of trivalent metal Schiff base complexes, J Serb Chem Soc, 75 (2010), 629‒637.
- R. R. Mohamed and A. M. Fekry, Antimicrobial and anticorrosive activity of adsorbents based on chitosan Schiff's base, Int J Electrochem Sci, 6 (2011), 2488‒2508.
- H. F. Abd El-Halim, M. M. Omar, G. G. Mohamed, and M. A. El-Ela Sayed, Spectroscopic and biological activity studies on tridentate Schiff base ligands and their transition metal complexes, Eur J Chem, 2 (2011), 178‒188.
- N. V. Tverdova, E. D. Pelevina, N. I. Giricheva, G. V. Girichev, N. P. Kuzmina, and O. V. Kotova, Molecular structures of 3d metal complexes with various Schiff bases studied by gas-phase electron diffraction and quantum-chemical calculations, J Mol Struct, 1012 (2012), 151‒161.
- C. Anitha, C. D. Sheela, P. Tharmaraj, and S. Sumathi, Spectroscopic studies and biological evaluation of some transition metal complexes of azo Schiff-base ligand derived from (1-phenyl-2,3-dimethyl-4-aminopyrazol-5-one) and 5-((4-chlorophenyl)diazenyl)-2-hydroxybenzaldehyde, Spectrochim Acta A Mol Biomol Spectrosc, 96 (2012), 493‒500.
- N. Raman, S. Sobha, and A. Thamaraichelvan, A novel bioactive tyramine derived Schiff base and its transition metal complexes as selective DNA binding agents, Spectrochim Acta A Mol Biomol Spectrosc, 78 (2011), 888‒898.
- R. A. A. Ammar and A. M. A. Alaghaz, Synthesis, spectroscopic characterization and potentiometric studies of a tetradentate [N2O2] Schiff base, N,N′-bis(2-hydroxybenzylidene)-1,1-diaminoethane and its Co(II), Ni(II), Cu(II) and Zn(II) complexes, Int J Electrochem Sci, 8 (2013), 8686‒8699.
- M. B. Halli, R. B. Sumathi, and M. Kinni, Synthesis, spectroscopic characterization and biological evaluation studies of Schiff's base derived from naphthofuran-2-carbohydrazide with 8-formyl-7-hydroxy-4-methyl coumarin and its metal complexes, Spectrochim Acta A Mol Biomol Spectrosc, 99 (2012), 46‒56.
- A. D. Garnovskii and I. S. Vasil′chenko, Rational design of metal coordination compounds with azomethine ligands, Russ Chem Rev, 71 (2002), 943‒996.
- J. L. Dean, Lange’s Handbook of Chemistry, McGraw-Hill, New York, 14th ed., 1992.
- A. A. Belal, M. A. Zayed, M. El-Desawy, and S. M. Rakha, Structure investigation of three hydrazones Schiff's bases by spectroscopic, thermal and molecular orbital calculations and their biological activities, Spectrochim Acta A Mol Biomol Spectrosc, 138 (2015), 49‒57.
- S. Ilhan, H. Temel, I. Yilmaz, and M. Sekerci, Synthesis, structural characterization and electrochemical studies of new macrocyclic Schiff base containing pyridine head and its metal complexes, J Organomet Chem, 692 (2007), 3855‒3865.
- E. M. Zayed, M. A. Zayed, and M. El-Desawy, Preparation and structure investigation of novel Schiff bases using spectroscopic, thermal analyses and molecular orbital calculations and studying their biological activities, Spectrochim Acta A Mol Biomol Spectrosc, 134 (2015), 155‒164.
- F. A. Cotton, G. Wilkinson, C. A. Murillo, and M. Bochmann, Advanced Inorganic Chemistry, John Wiley & Sons, New York, 6th ed., 1999.
- E. M. Zayed and M. A. Zayed, Synthesis of novel Schiff's bases of highly potential biological activities and their structure investigation, Spectrochim Acta A Mol Biomol Spectrosc, 143 (2015), 81‒90.
- M. A. Zayed, M. A. Fahmey, M. El-Desawy, and Y. S. Farrag, Structure characterization of terazosin drug using mass spectrometry and thermal analyses techniques in comparison with semi-empirical molecular orbital (MO) calculations, J Therm Anal Calorim, 120 (2015), 1061‒1069.
- M. Karthikeyan, D. Prasad, B. Poojary, K. Subrahmanya Bhat, B. Holla, and N. Kumari, Synthesis and biological activity of Schiff and Mannich bases bearing 2,4-dichloro-5-fluorophenyl moiety, Bioorg Med Chem, 14 (2006), 7482‒7489.
- N. Shahabadi, Z. Ghasemian, and S. Hadidi, Binding studies of a new water-soluble iron(III) Schiff base complex to DNA using multispectroscopic methods, Bioinorg Chem Appl, 2012 (2012), 126451.
- S. Sen, N. A. Farooqui, S. Dutta, T. S. Easwari, V. Gangwar, K. Upadhya, et al., Physicochemical and biological evaluation of some Schiff base by conventional and microwave assisted method, Der Pharma Chemica, 5 (2013), 128‒134.
- K. Singh, M. S. Barwa, and P. Tyagi, Synthesis, characterization and biological studies of Co(II), Ni(II), Cu(II) and Zn(II) complexes with bidentate Schiff bases derived by heterocyclic ketone, Eur J Med Chem, 41 (2006), 147‒153.
- M. M. H. Khalil, E. H. Ismail, G. G. Mohamed, E. M. Zayed, and A. Badr, Synthesis and characterization of a novel Schiff base metal complexes and their application in determination of iron in different types of natural water, Open J Inorg Chem, 2 (2012), 18646.
- A. M. Hartman, M. Mondal, N. Radeva, G. Klebe, and A. K. H. Hirsch, Structure-based optimization of inhibitors of the aspartic protease endothiapepsin, Int J Mol Sci, 16 (2015), 19184‒19194.
- M. B. Rao, A. M. Tanksale, M. S. Ghatge, and V. V. Deshpande, Molecular and biotechnological aspects of microbial proteases, Microbiol Mol Biol Rev, 62 (1998), 597‒635.
- J. R. Naglik, S. J. Challacombe, and B. Hube, Candida albicans secreted aspartyl proteinases in virulence and pathogenesis, Microbiol Mol Biol Rev, 67 (2003), 400‒428.
- P. K. Vuppala, D. R. Janagam, and P. Balabathula, Importance of ADME and bioanalysis in the drug discovery, J Bioequiv Availab, 5 (2013), e31.
- R. O. Potts and R. H. Guy, Predicting skin permeability, Pharm Res, 9 (1992), 663‒669.
- J. B. Baell and G. A. Holloway, New substructure filters for removal of pan assay interference compounds (pains) from screening libraries and for their exclusion in bioassays, J Med Chem, 53 (2010), 2719‒2740.
Copyright © 2018 M. A. Zayed et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.